Case Study: Using a fluorescence spectroscopy method to determine the cell density of the cyanobacteria Anabaena flos-aquae

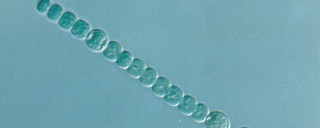
Anabaena flos-aqaue is a species of filamentous cyanobacteria used in ecotoxicology testing.
It forms long chains of cells which makes some methods of cell density determination either extremely laborious or impractical. e.g. counting using a microscope with a haemocytometer slide or automated particle counters (Coulter counter).
Approach:
Literature showed that using a fluorescence spectrometer to measure cyanobacteria or algal cell density was a reliable and accurate method. The absorption of fluorescence by the fluorophore phycocyanin (a fluorescent blue pigment protein found in cyanobacteria and some algae) was found to be a suitable surrogate to more direct cell counting methods. There was data readily available on the excitation and emission wavelengths for phycocyanin which we used to run growth trials and a reference toxicant study, where a chemical with a known toxicity level is used to test the efficacy and effectiveness of a test method. The test was run for 72 hours with samples taken at each 24-hour interval for cell density determination.


Outcome: After some trial and error, we were able to successfully determine the cell density of test samples at the 24-, 48- and 72-hour sampling occasions and meet the validity criteria detailed in an internationally recognised test guideline (OECD 201, 2011) for several regulatory studies.
It quickly became clear that the method worked well for the 48-and 72-hour sampling occasions but due to the relatively low concentration of cells at the 24-hour time point, measured values were not always discernible from the background readings and were not within our planned calibration range. It was thought that the filamentous nature of A. flos-aquae contributed to this as it was difficult to obtain a homogenous sample. Disruption of the cell chains using an ultrasonic bath was undertaken, leading to shorter chains comprised of fewer cells, increasing the homogeneity of the samples. Alongside this, we tested increasing the initial cell density of A. flos-aquae inoculated into each test vessel at the start of the test at several different levels to determine what was most effective – able to grow exponentially over the whole test period and achieve the highest cell density at the 24-hour timepoint. Having increased the homogeneity of our samples, we were also able to extend our calibration range to cover much lower cell densities.
Past Poster Presentations & Case Studies



This website uses cookies to ensure you get the best experience. Learn more